November 16, 2019
Bangkok, Thailand
ISBT Asia
ISBT Asia, November 16 - 19 2019
Fresenius Medical Care, the world’s largest provider of dialysis services and products, celebrated today the 40th anniversary of the company’s Schweinfurt, Germany plant. Joining employees and their family members for the festivities were numerous business and political representatives, including Schweinfurt Mayor Sebastian Remelé.
Schweinfurt is Fresenius Medical Care’s largest development and manufacturing facility for dialysis machines and other medical technology devices. Over the past four decades more than 800,000 dialysis machines have been made at the site, where the company is now investing some €20 million in a new, 8,000-square-meter (86,000-square-foot) technology center. The Schweinfurt facility’s workforce now numbers more than 1,200, with about a third of the employees involved in research and development.
“Schweinfurt is a huge success story – not just for Fresenius Medical Care, but even more so for our patients around the world,” said Rice Powell, CEO of Fresenius Medical Care. “For 40 years now, we have been developing life-sustaining products here at the very highest level, and we’re doing our best to continue writing new chapters in that success story. With the building of the new technology center, we are working to mesh development and production at this site even closer together, so that we can provide even better dialysis machines for our patients in the future.”
“I am very proud that Fresenius Medical Care is established in Schweinfurt and from here developed into the world market leader for dialysis machines,” said Mayor Remelé. “These dialysis systems are used to treat several hundred thousand patients with chronic kidney failure. In the name of the city of Schweinfurt, and personally, I want to wish Fresenius Medical Care continued great success in this important field.”
The plant’s history dates back to 1979 and the acquisition of a vacant factory building in a Schweinfurt industrial zone. There, under the business name MTS Medizin-Technische Systeme Schweinfurt GmbH, 40 employees began producing the first dialysis machine developed by Fresenius – the A2008C, which won the gold medal for technical innovation at the Leipzig Trade Fair that same year. By 1984, every second dialysis machine produced in Germany was “Made in Schweinfurt.” The A2008C would go on to become the world’s leading and best-selling dialysis machine, a position Fresenius Medical Care still holds following the introduction of the A2008C’s successor models. Today, more than half of all dialysis machines sold worldwide are made by Fresenius Medical Care.
Dialysis machines, bloodline systems and dialyzers – the latter often dubbed “artificial kidneys” because this is where the blood is cleaned – are the most important products for treating chronic kidney disease. During treatment, the dialysis machine pumps the patient’s blood through bloodlines, monitors its circulation through the dialyzer, and adds anti-coagulants. Treatments are generally carried out three times weekly, taking between three and six hours each.
This release contains forward-looking statements that are subject to various risks and uncertainties. Actual results could differ materially from those described in these forward-looking statements due to certain factors, including changes in business, economic and competitive conditions, regulatory reforms, foreign exchange rate fluctuations, uncertainties in litigation or investigative proceedings, and the availability of financing. These and other risks and uncertainties are detailed in Fresenius Medical Care AG & Co. KGaA's reports filed with the U.S. Securities and Exchange Commission. Fresenius Medical Care AG & Co. KGaA does not undertake any responsibility to update the forward-looking statements in this release.
Fresenius Medical Care, the world’s largest provider of dialysis products and services, is pleased the U.S. administration’s plans for changing the way care is provided to people with kidney disease supports its existing strategy. The company has long worked on various initiatives to promote home dialysis, improve access to transplants, and develop new, value-based care models for chronic kidney disease patients.
Rice Powell, CEO of Fresenius Medical Care, said: “We congratulate the Administration on today’s announcement and celebrate the proposed initiatives as a win for our patients and for the 30 million Americans living with kidney disease. We are committed to continuously improving the quality of life of patients affected by kidney disease and have already established initiatives to improve prevention, offer more flexible treatment options, introduce value-based care models and promote organ donation –in the United States and abroad.
We share the U.S. Administration’s commitment to expanding access to home dialysis, transplantation and new models of value-based care for chronic kidney disease, and we see it as an endorsement of our initiatives. We invest constantly in innovation and will continue to do so in order to further develop the healthcare system. The proposed reimbursement models and new incentives will help foster further innovation and support a healthcare delivery system structure that is closely attuned to the needs of our patients.
Our recent merger with NxStage, which makes the leading hemodialysis machine for home use, is just one piece of a focused effort to educate patients and physicians around the benefits of home treatment and provide industry leading solutions to enable them to do so. We are also investing in technologies for the future, including new innovations for remote patient monitoring and telehealth that, combined with predictive analytics and artificial intelligence, will make it easier to help patients between visits to a doctor and avoid unnecessary hospitalizations.
We welcome reimbursement reforms that facilitate investments in care models designed to improve outcomes and help reduce costs, two goals to which more use of home dialysis and transplants can equally contribute. We will carefully review the U.S. administration’s proposal and contribute to developing the framework that offers the best possible conditions and greatest benefit for patients.”
Fresenius Medical Care's Global Medical Office conducts research in various fields of prevention and the use of clinical data to develop optimal treatment paths while avoiding unnecessary and expensive complications.
The company is also active in the field of regenerative medicine, and is a pioneer in testing flat-rate and value-based reimbursement models. The End Stage Renal Disease Seamless Care Organizations (ESCOs) of Fresenius Medical Care, which were established in close cooperation with the U.S. Centers for Medicare & Medicaid Services (CMS), have already achieved improved treatment outcomes and cost savings.
This release contains forward-looking statements that are subject to various risks and uncertainties. Actual results could differ materially from those described in these forward-looking statements due to certain factors, including changes in business, economic and competitive conditions, regulatory reforms, foreign exchange rate fluctuations, uncertainties in litigation or investigative proceedings, and the availability of financing. These and other risks and uncertainties are detailed in Fresenius Medical Care AG & Co. KGaA's reports filed with the U.S. Securities and Exchange Commission. Fresenius Medical Care AG & Co. KGaA does not undertake any responsibility to update the forward-looking statements in this release.
Fresenius Medical Care North America (FMCNA) has announced an investment in BioIntelliSense, a Denver-based company that is developing a medical grade data services platform for continuous remote health monitoring, predictive analytics and algorithmic clinical insights. FMCNA is committed to bringing its expertise in advanced analytics and artificial intelligence to the development and deployment of this system in cooperation with BioIntelliSense. The aim is to jointly create clinical pathways that can alert clinicians to the need for early intervention, potentially preventing patients from complications and, thus, reducing unnecessary as well as costly hospitalizations. The investment, therefore, supports Fresenius Medical Care’s strategic goal to improve monitoring, interventions and outcomes for patients living with kidney disease and other chronic illnesses in order to increase their quality of life.
Fresenius Kabi launches Levothyroxine Sodium Injection in the U.S. It is the first, and only, pre-mixed solution of this medication currently available in the U.S.
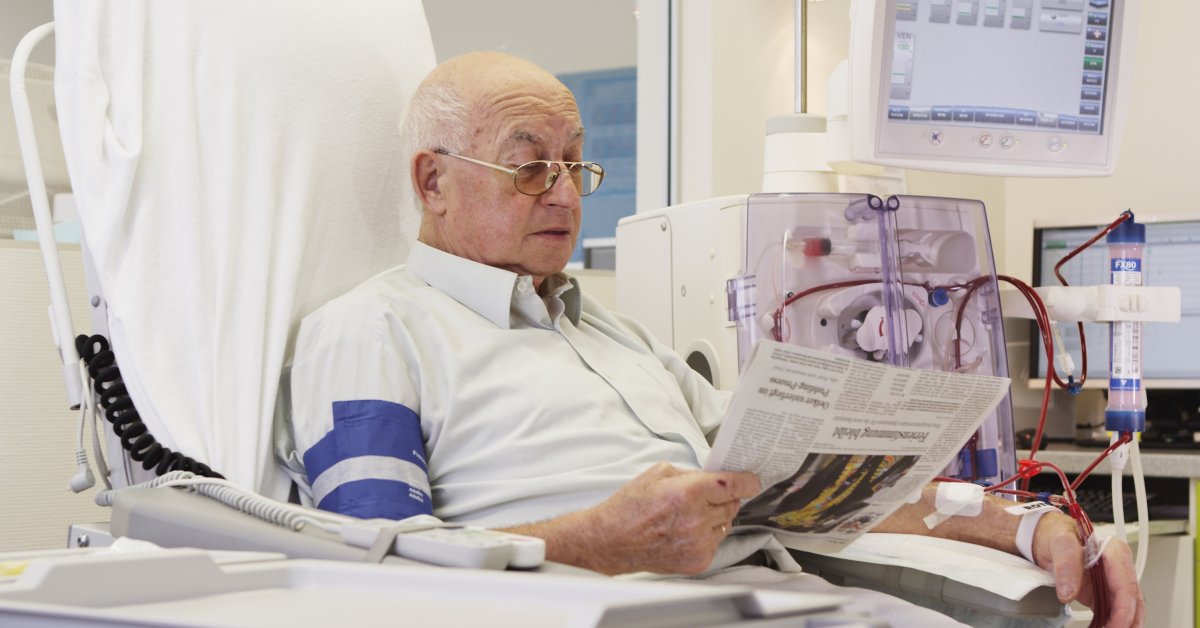
Fresenius Medical Care, the world’s largest provider of dialysis products and services, is expanding its clinical research activities. Frenova Renal Research (Frenova), previously a Fresenius Medical Care North America subsidiary, will now offer its services worldwide and will be integrated into the new Global Medical Office headed by Dr. Frank Maddux, Chief Medical Officer.
Frenova offers services for the clinical development of medicines and medical products in the field of kidney research. This offering, previously limited to North America, will now be linked with the corresponding services of Fresenius Medical Care’s Europe, Middle East and Africa (EMEA) and Latin America regions and bundled under Frenova. This will enable the company to draw on a network of more than 550 researchers at over 350 locations.
“Frenova is active at the intersection of clinical research and patient care,” said Dr. Maddux. “No other provider of clinical development services understands the medical needs of people living with kidney disease as well as we do. By making this expertise available worldwide, we will enable a faster, more efficient development of medications and other products. This is an additional, important building block for improving their quality of life.”
Disclaimer
This release contains forward-looking statements that are subject to various risks and uncertainties. Actual results could differ materially from those described in these forward-looking statements due to certain factors, including changes in business, economic and competitive conditions, regulatory reforms, foreign exchange rate fluctuations, uncertainties in litigation or investigative proceedings, and the availability of financing. These and other risks and uncertainties are detailed in Fresenius Medical Care AG & Co. KGaA's reports filed with the U.S. Securities and Exchange Commission. Fresenius Medical Care AG & Co. KGaA does not undertake any responsibility to update the forward-looking statements in this release.
Dexmedetomidine Hydrochloride in 0.9% Sodium Chloride Injection is the latest addition to the company´s growing anesthesia and analgesia portfolio.
June 28, 2019
Amsterdam, The Netherlands
Kepler Cheuvreux One Stop Shop Amsterdam
Fresenius Medical Care, the world’s largest provider of dialysis products and services, yesterday successfully placed bonds with an aggregate principal amount of USD 500 million. The bonds have a maturity of 10 years and an annual coupon of 3.750%. The issue price is 98.461%, resulting in a yield of 3.938%.
The proceeds will be used for general corporate purposes, including the refinancing of outstanding indebtedness.
Application will be made for admission to trading of the bonds on the regulated market of the Luxembourg Stock Exchange.
Disclaimer
This announcement does not contain or constitute an offer of, or the solicitation of an offer to buy or subscribe for, securities to any person in Australia, Canada, Japan, or the United States of America (the “United States”) or in any jurisdiction to whom or in which such offer or solicitation is unlawful. The securities referred to herein have not been and will not be registered under U.S. Securities Act of 1933, as amended (the “Securities Act”) and may not be offered or sold in the United States or to, or for the account or benefit of, U.S. persons, absent such registration, except pursuant to an exemption from, or in a transaction not subject to, the registration requirements of the Securities Act. Subject to certain exceptions, the securities referred to herein may not be offered or sold in Australia, Canada or Japan or to, or for the account or benefit of, any national, resident or citizen of Australia, Canada or Japan. The offer and sale of the securities referred to herein has not been and will not be registered under the Securities Act or under the applicable securities laws of Australia, Canada or Japan. There will be no public offer of the securities in the United States.
This announcement has been prepared on the basis that any offer of Notes in any Member State of the European Economic Area (EEA) which has implemented the Prospectus Directive (2003/71/EC), as amended (each, a Relevant Member State) will be made pursuant to an exemption under the Prospectus Directive, as implemented in that Relevant Member State, from the requirement to publish a prospectus for offers of securities. Fresenius Medical Care AG & Co. KGaA has not authorized, nor does it authorize, the making of any offer of securities in circumstances in which an obligation arises for Fresenius Medical Care AG & Co. KGaA or any other person to publish or supplement a prospectus for such offer.
This announcement is directed at and/or for distribution in the United Kingdom only to (i) persons who have professional experience in matters relating to investments falling within article 19(5) of the Financial Services and Markets Act 2000 (Financial Promotion) Order 2005 (the “Order”) or (ii) high net worth entities falling within article 49(2)(a) to (d) of the Order (all such persons are referred to herein as “relevant persons”). This announcement is directed only at relevant persons. Any person who is not a relevant person should not act or rely on this announcement or any of its contents. Any investment or investment activity to which this announcement relates is available only to relevant persons and will be engaged in only with relevant persons.
This announcement contains forward-looking statements that are subject to various risks and uncertainties. Future results could differ materially from those described in these forward-looking statements due to certain factors, e.g. changes in business, economic and competitive conditions, regulatory reforms, results of clinical trials, foreign exchange rate fluctuations, uncertainties in litigation or investigative proceedings, and the availability of financing. Fresenius Medical Care AG & Co. KGaA does not undertake any responsibility to update the forward-looking statements in this announcement. In furnishing our website address in this announcement, we do not intend to incorporate any information on our website into this announcement.
Neostigmine is now available in the company’s Simplist® ready-to-administer prefilled syringes, expanding the company´s analgesia and anesthesia portfolio.








