Fresenius Medical Care, the world’s largest provider of dialysis products and services, today announced the closing of the divestiture of Sound Inpatient Physicians Holdings, LLC (Sound) to an investment consortium led by Summit Partners as of June 28, 2018. The total transaction proceeds are $2.15 billion (EUR 1.85 billion1).
The divestment of Sound is aligned with Fresenius Medical Care’s goal of further sharpening the profile of the company’s Care Coordination portfolio. The transaction is expected to generate a positive effect on net income2 for Fresenius Medical Care of around USD 7523 million (EUR 648 million1). This effect includes the half year impact from the increase in valuation of Sound´s share based payment program caused by the divestment of Sound.
The financial targets for 2018 and 2020 do not include the effects of this divestiture. The divestment reduces on a constant currency basis Fresenius Medical Care´s 2018 revenue expectations by around EUR 650 million and net income2 by around EUR 40 million. The growth rates indicated in the financial targets for 2018 will be achieved on a comparable basis4 following the divestiture of Sound.
1 EUR/USD 1.16
2 Net income attributable to shareholders of Fresenius Medical Care AG & Co. KGaA
3 Based on the company´s latest available information
4 On a pro-forma basis revenue for Sound in H2 2017 was around EUR 560 million and net income was around EUR 40 million
This release contains forward-looking statements that are subject to various risks and uncertainties. Actual results could differ materially from those described in these forward-looking statements due to certain factors, including changes in business, economic and competitive conditions, regulatory reforms, foreign exchange rate fluctuations, uncertainties in litigation or investigative proceedings, and the availability of financing. These and other risks and uncertainties are detailed in Fresenius Medical Care AG & Co. KGaA's reports filed with the U.S. Securities and Exchange Commission. Fresenius Medical Care AG & Co. KGaA does not undertake any responsibility to update the forward-looking statements in this release.
Fresenius Kabi is expanding its production and warehouse capacity in Runcorn, England, to meet increasing demand in United Kingdom’s homecare market and from UK and Ireland hospitals. The expansion’s first stage will be a new distribution and operations center, scheduled to open in early 2019. A second services unit for producing aseptically compounded parenteral nutrition products will be completed by 2020. Fresenius Kabi will invest over €9 million in the expansion.
Fresenius Kabi is expanding its production and warehouse capacity in Runcorn, England, to meet increasing demand in United Kingdom’s homecare market and from UK and Ireland hospitals. The expansion’s first stage will be a new distribution and operations center, scheduled to open in early 2019. A second services unit for producing aseptically compounded parenteral nutrition products will be completed by 2020. Fresenius Kabi will invest over €9 million in the expansion.
June 27, 2018
Zurich, Switzerland
Credit Suisse – European Medtech & Healthcare Services Day
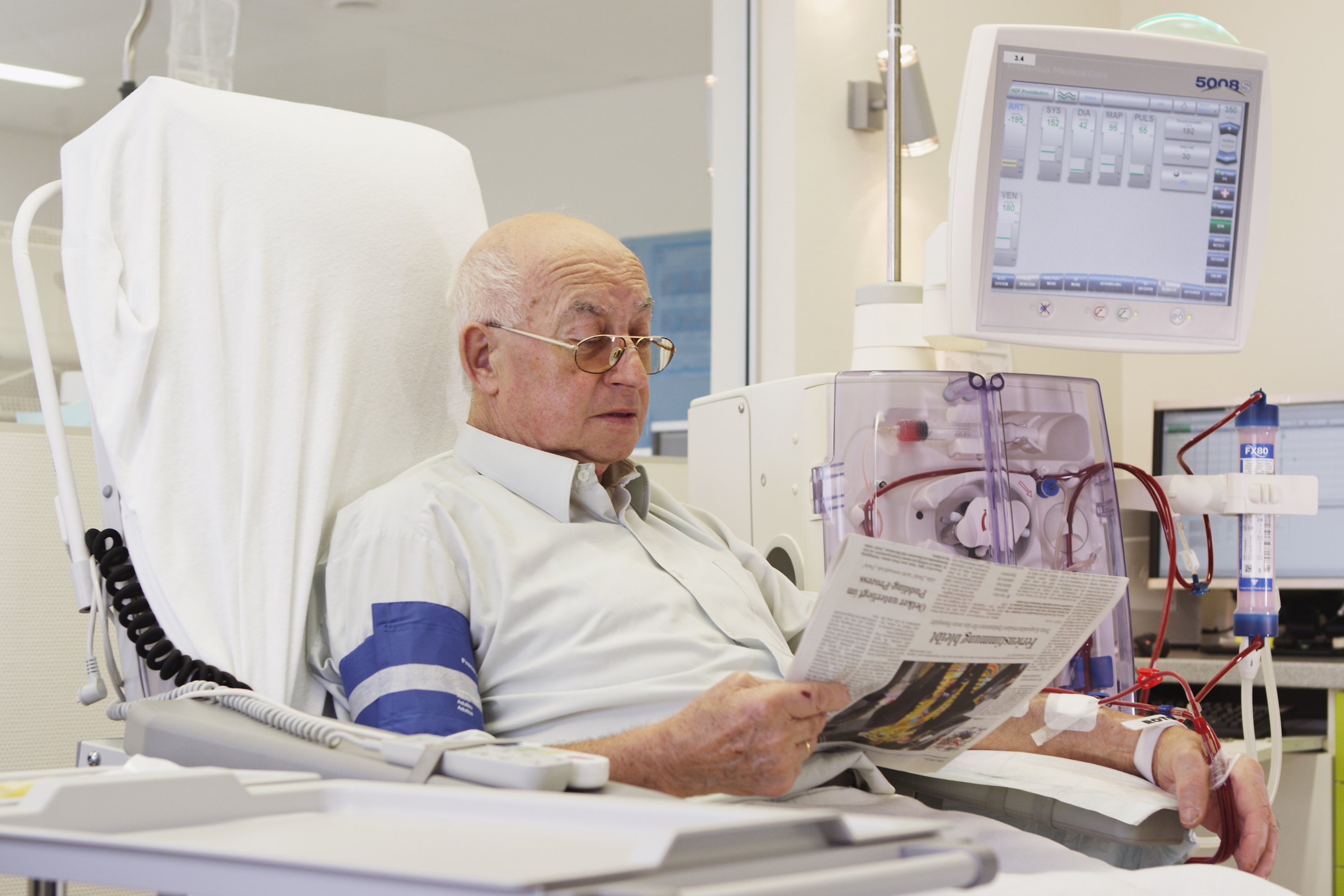
Fresenius Medical Care, the world’s largest provider of dialysis products and services, and Humacyte, Inc., a medical research, discovery and development company, today announced a strategic, global partnership and a $150M USD equity investment. This agreement has the potential to make Humacyte’s investigational human acellular vessel, HUMACYL®, available to more patients worldwide following approval of the product. HUMACYL is currently being investigated for vascular access for hemodialysis and may prove more effective than current synthetic grafts and fistula. Under the terms of the agreement, Fresenius Medical Care will obtain the exclusive global rights to commercialize HUMACYL.
Fresenius Medical Care will be responsible for the marketing, sales and distribution of HUMACYL following approval by the relevant health authorities. In addition, Fresenius Medical Care will make a $150M USD equity investment in Humacyte to gain a 19% fully diluted ownership stake in the company. With the investment, Fresenius Medical Care will have the opportunity to bring transformative clinical innovation in the form of Humacyte’s bioengineered human acellular vessels to the worldwide end stage renal disease (ESRD) patient population following product approval. The transaction is subject to customary closing conditions, including expiration or termination of the waiting period under the Hart-Scott-Rodino Antitrust Improvements Act of 1976, as amended, and is expected to close in July 2018.
“By partnering with Humacyte, Fresenius Medical Care has an opportunity to offer a dialysis vascular access option with the potential for significant clinical efficacy and safety improvements, including the potential to minimize catheter contact time to the benefit of our patients,” said Franklin Maddux, MD, Chief Medical Officer for Fresenius Medical Care North America. “Our exclusive rights to distribute this innovative technology to dialysis patients worldwide may have significant benefits not only to patients, but health systems as well. With the potential for fewer anticipated complications and interventions compared to synthetic grafts, we may see increased safety for patients and reduced medical and economic burdens to the healthcare system.”
The current vascular access modalities necessary to deliver dialysis treatment include fistulas, grafts and central venous catheters. All three options have limitations. Half of fistulas fail and do not mature in patients, delaying vascular access for dialysis treatment. In the meantime, many patients need a central venous catheter, which significantly increases the risk of infection. Humacyte has developed a novel human tissue-based investigational product, HUMACYL, for patients with ESRD requiring hemodialysis. Compared to an arteriovenous fistula, HUMACYL can be available for use in hemodialysis within weeks and may have an overall higher rate of maturation. It also may offer a more durable, biologic alternative to synthetic grafts.
“This is a transformational milestone for Humacyte, giving us the world’s strongest partner to help bring our product to more patients globally,” said Carrie Cox, CEO and Chairman of Humacyte, Inc. “Our partnership will allow Humacyte to focus on advancing the potential for HUMACYL as a substantial breakthrough in the science of regenerative medicine, and to continue our development of an exciting future pipeline.”
Humacyte’s bioengineered blood vessel is currently in Phase III pivotal trials in the U.S. and Europe, and the company plans to seek regulatory approval in both regions upon completion of the trials.
Disclaimer
This release contains forward-looking statements that are subject to various risks and uncertainties. Actual results could differ materially from those described in these forward-looking statements due to certain factors, including changes in business, economic and competitive conditions, risks and uncertainties in research and development and the regulatory approval process; failure to satisfy the conditions to the consummation of the transaction, including the receipt of regulatory approval; regulatory reforms, foreign exchange rate fluctuations, uncertainties in litigation or investigative proceedings, and the availability of financing. These and other risks and uncertainties are detailed in Fresenius Medical Care AG & Co. KGaA's reports filed with the U.S. Securities and Exchange Commission. Without prejudice to its obligations under capital market laws, neither Fresenius Medical Care AG & Co. KGaA nor Humacyte, Inc. undertakes any responsibility to update the forward-looking statements in this release.
June 12, 2018
Rancho Palos Verdes, USA
Goldman Sachs 39th Annual Global Healthcare Conference
June 12 – 13, 2018
More than a year after closing its acquisition of the Spanish hospital operator Quirónsalud, Fresenius Helios foresees strong prospects for further international growth. At a Capital Markets Day in Berlin today, Fresenius provided investors and analysts with an overview of the progress made in the cooperation between Helios Germany and Quirónsalud, which is opening up new growth opportunities in many areas.
Fresenius Helios confirmed the medium-term target for synergies of €50 million annually. Cooperating in laboratory services and joint purchasing has already achieved cost savings in the current business year that should increase to €30 million per year in the medium term. Annual sales synergies of about €20 million are expected from knowledge transfers in medicine, new models for patient care, and digitalization, among other measures.
Quirónsalud has already begun to implement the system developed by Helios Germany for measuring and assessing data on medical quality. To increase transparency and spur individual hospitals to compete on quality, Helios Germany publishes treatment data on the most important and common medical indications for each hospital in direct comparison with national averages compiled by Germany’s Federal Statistical Office. Quirónsalud has already adapted most of these quality indicators. It has also launched peer reviews, collegial exchanges in which the responsible physicians from the individual hospitals advise and consult with each other on questions concerning treatment quality. These exchanges have already led to significant quality improvements at Helios Germany.
The knowledge transfer also extends to digitalization: Quirónsalud’s advanced know-how in patient-oriented uses includes the development of apps, while Helios Germany is particularly strong in IT processes.
Stephan Sturm, CEO of Fresenius, said: “Helios Germany and Quirónsalud are bundling their respective strengths across national borders, exchanging experience and knowledge. This benefits our patients, in Spain as well as in Germany. And it is creating, step by step, the economic prerequisites for the further internationalization of our hospital business. The driving force behind our business success is, and will remain, our clear focus on the well-being of patients. So wherever and whenever we can do more for our patients through closer cooperation between our business segments, we will seize that opportunity.”
Fresenius Helios will also profit from combining the experience gained from the very different health care systems of Spain and Germany. The Spanish system, for its part, allows a great deal of flexibility in care provision models. In Madrid, for example, Quirónsalud has been given responsibility for providing health care to publicly funded patients in designated parts of the city in return for a set reimbursement rate. For patients, however, treatment quality remains the decisive criterion, and assigned patients are still free to choose another hospital. If they do, the cost must be assumed by the hospital that was originally assigned to their care. This increased competition stimulates continuous improvement in areas that are of crucial importance to patients – such as medical quality, service and shorter waiting times. All of these are core competencies of Quirónsalud.
Another area where Fresenius Helios can now draw on its experience from two different health care systems is in classifications of inpatient and outpatient care. The two are strictly separated in Germany, but less so in Spain: Many treatments and after-care procedures that Germans undergo as inpatients are provided to their Spanish counterparts on an outpatient basis – often resulting in significantly shorter hospital stays.
Dr. Francesco de Meo, who is responsible for Fresenius Helios on the Fresenius Management Board, said: “Helios Germany and Quirónsalud are leaders in their home markets, each of which has different reimbursement and care models and health insurance systems. In response, each company has developed its own strengths, which excellently complement each other and can be used to their mutual advantage. We expect this will contribute to higher medical quality and more efficiency, and bring us closer to patients. Thus, together we are building a common base of knowledge and experience that will help us to enter new markets.”
Cooperation with Fresenius Vamed is also being intensified – for example, in procurement, where Fresenius Helios and Fresenius Vamed are now jointly purchasing certain products. In addition, Fresenius Vamed has started providing Quirónsalud hospitals in Spain with technical services and medical technology, as it was already doing for Helios Germany. In Germany, meanwhile, Fresenius Helios and Fresenius Vamed will combine their know-how in hospital construction in order to bundle their competencies in construction and project management.
Earlier this week, the two companies agreed that Fresenius Helios’ inpatient rehabilitation business in Germany will be transferred to Fresenius Vamed on July 1, 2018. This will put Fresenius Helios on a stronger growth footing, with an even clearer focus on the acute-care hospital business and its further internationalization.
Webcast of the event:
Fresenius Helios Capital Markets Day will be available as a webcast on the Internet under: www.fresenius.com/investors-new-developments
This release contains forward-looking statements that are subject to various risks and uncertainties. Future results could differ materially from those described in these forward-looking statements due to certain factors, e.g. changes in business, economic and competitive conditions, regulatory reforms, results of clinical trials, foreign exchange rate fluctuations, uncertainties in litigation or investigative proceedings, and the availability of financing. Fresenius does not undertake any responsibility to update the forward-looking statements in this release.



