
When the spine is operated on, mere millimeters can be critical. For such highly complex procedures, surgeons need sound judgment, precise spatial awareness, the capacity for abstraction – and a skilled pair of hands. Yet by their very nature, humans frequently reach the limits of their abilities. That is why many hospitals have for a long time been using high-resolution three-dimensional images and surgical navigation systems, such as spinal navigation, to perform their work. But all of this is only the start.
(Published: August 2025)
The Department of Neurosurgery and the Center for Spine Therapy at Helios Hospital Berlin-Buch have continued to build on their expertise in spinal surgical procedures: Under the direction of chief physician Professor Yu-Mi Ryang, the department has been using augmented reality (AR) in spinal stabilization surgery for the past two years. Berlin-Buch is thus one of the first hospitals in Germany to use AI in neurosurgery for the benefit of patients. The AR technology makes the procedures more precise, less invasive, and also shorter. A look over the shoulder of Prof. Ryang reveals exactly how AR is used here and why this represents an important step into the future.

There is total concentration in the operation room. Prof. Ryang is leaning over her patient. He is suffering from a spinal tumor that has already partially destroyed the bones and is also pressing on the highly sensitive spinal cord. Over the course of the next two to three hours, the surgeon will remove the tumor from the spinal cord and strengthen the area of the spine destabilized by the tumor using a so-called screw and rod system. It should restabilize the spine and, above all else, free the patient from pain. The neurosurgery teams also use such minimally invasive techniques for other indications, combining them with AR technology – in frequently occurring cases of wear and tear of the spine, for example, as well as when repairing fractured vertebrae after an accident or treating osteoporosis.
In purely visual terms, this screw and rod system resembles a railroad track. Firstly, between four and eight, sometimes significantly more, screws are firmly anchored in the “track bed” – i.e. the vertebral bodies. To this end, the screws are attached to so-called towers, which function as a kind of screw extension. Without these towers, the screws would disappear into the depth of the surgical area when inserted. They would no longer be visible to the surgeon and would hardly be accessible through the back muscles.
All of this is preferably performed in a minimally invasive procedure by making small incisions in the skin in order to preserve the tissue and reduce blood loss. Other advantages of this tissue-preserving procedure include a shorter operating time. This is because there is no longer any need for the complex process of detaching the back muscles from the spine, as is the case with open procedures. The patient also suffers less postoperative pain.
Due to the minimally invasive technique, Prof. Ryang may only be able to see a small section of the surgical area with her own eyes, but this is no obstacle thanks to AI. The precise location where the rods have to be inserted and, above all, the required length and curvature, along with the location of other important structures of the spine – such as the spinal cord or the tumor itself – can all be shown to her with the help of augmented reality. The latter also increases the safety of the procedure.

As the operation progresses, things get tricky again: Depending on the curvature of the spine, Prof. Ryang has to shape the titanium rods in advance so that they fit perfectly into the screw heads. Being even a few millimeters out can make a big difference here. In the past, the team relied purely on operative experience and their own judgment when shaping and fitting the rods. “This can be extremely difficult, however, particularly with severe curvatures of the spine,” explains the surgeon. Furthermore, it is relatively time-consuming. This is precisely where AR can now help: “With the aid of augmented reality, we can determine the ideal position, length, and curvature of the rods during the ongoing operation in a matter of minutes and prepare them accordingly.”
When the term augmented reality (AR) is mentioned, many people tend to think of virtual reality (VR) headsets. In Berlin-Buch, however, the neurosurgery team works with standard tablets equipped with a built-in camera and special software. Dr. Ryang points the tablet camera at the patient’s back. By swiveling the tablet, the surgical area is scanned – and with it the QR codes affixed to the screw tower in advance. The virtual 3D towers and virtual rods then gradually appear on the tablet, which are assigned to the real-time image of the patient. “Changes in position during the procedure are also no problem for me – orientation is guaranteed at all times,” says Prof. Ryang. With the aid of the QR codes, the tablet’s integrated camera can determine the exact position of the inserted screws and the exact length and curvature of the rods. And it can “merge” with the patient’s real-time image as a virtual 3D image. Based on this augmented reality, the surgeon can then use the tablet to create custom rods for each individual patient.
The software works out a kind of template for this, enabling the surgeon to bend the titanium rods into the ideal shape by hand. To allow her to compare, she repeatedly places the rods on the template. In the past, if the rod was not perfectly bent, a screw could be ripped out of its position again after being inserted. The doctor says that this risk is now minimal: “We hope that there will be significantly fewer postoperative complications in the long term, such as a broken rod, a loose screw, or some other material failure. Any of these things would necessitate revision surgery. We also hope that this method will improve the clinical outcome of patients in terms of pain and quality of life.”
The neurosurgery team in Berlin-Buch has been using AR technology for about two years now. At the present time, the interim assessment looks promising: Several studies have shown that it improves operative performance. “Even highly experienced surgeons are not always able to set the ideal curvature radii of the screw and rod systems without augmented reality support,” says Dr. Ryang. “But with the help of AR, practically all surgeons are able to achieve perfect results.”
Yet it gets even better with augmented reality: AR-supported procedures lead to shorter operating times, thereby also reducing the length of time the patient spends under anesthetic. This represents a continuation of the positive trend seen in spinal treatments, with minimally invasive neurosurgical procedures on the back becoming more and more comfortable for the patient.
“Our example from Berlin-Buch shows what it means to bring a medical innovation to patients in a very practical way,” says a delighted Prof. Ryang. “It is not the one major revolution that changes everything overnight. Rather, we combine various innovative solutions to achieve better outcomes for our patients step by step,” says the expert in summary.
Top specialist in neurosurgery
Prof. Yu-Mi Ryang studied at Ruhr University Bochum, where she also gained her doctorate. The Düsseldorf-born physician then worked in the field of neurosurgery at University Hospital RWTH Aachen, where she completed her specialist training. Prof. Ryang attained her professorship at the Klinikum rechts der Isar of the Technical University of Munich and was appointed senior physician and deputy director of the Neurosurgical Department and Polyclinic in 2017. The specialist joined Fresenius Helios in Berlin-Buch in 2019. As chief physician, she heads the Neurosurgical Department as well as the Center for Spine Therapy.
She also holds a number of positions in various national and international professional societies, including the German Society of Neurosurgery, or DGNC, where Prof. Ryang is currently head of the spine section. In addition, she is on the board of the German Spine Society (DWG), where she serves on various commissions and is chairwoman of Module 5 of the DWG’s basic courses. In EUROSPINE, Prof. Ryang is also chairwoman of Module 4, and in AO Spine she is a member of the Technical Commission and the “Fracture, Tumor, Deformity Expert Group.”
(Published: March 2025)
The expert committee is made up of four international specialists from the fields of science, business, and consulting. It was established by Michael Moser, member of the Management Board, and helps Fresenius to further develop its sustainability strategy. Yesterday’s meeting focused on key issues such as water management and the ongoing development of the Group-wide human rights program.
Sarah Tix, Head of Group Sustainability, and Anahita Thoms, co-chairs of the Advisory Board, took time out from a tour of the plant to explain the importance of sustainability issues - start the video on the right.
Fresenius Kabi’s Friedberg site supplies essential medical devices used in the care of critically and chronically ill patients in more than 100 countries. Around 650 employees work at the plant. Every year, more than 125 million infusion solutions and around 70 million liquid medicines such as analgesics and narcotic agents are manufactured here. The adjacent logistics center puts together around 40,000 consignments every month and is responsible for packing and shipping them.
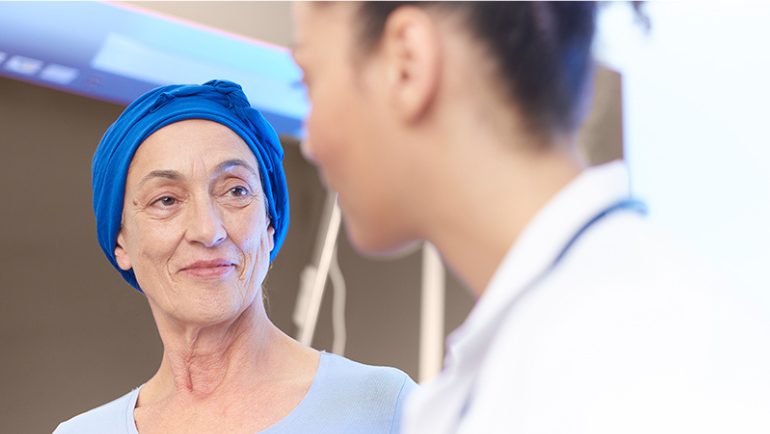
“I have cancer.” Each year, around half a million people in Germany alone have to utter these difficult words. The diagnosis casts a shadow over the lives of patients and their relatives. There are many fears and worries, but also hope and a sense of togetherness – all these emotions connect those affected. And yet each patient and each illness is unique.
(Published: April 2025)
This year’s World Cancer Day highlights the commitment to modern cancer care under the theme “United by Unique.” It is a kind of care that focuses not just on the disease, but on the people, taking a holistic approach to their treatment.
Fresenius is also fully committed to this approach. Our employees support individuals affected by cancer in all phases of the disease: from prevention and the initial diagnosis to therapy and home care. They serve patients beyond expectations and always give their best – not just in the fight against cancer, but also when striving for the highest possible quality of life for patients. Completely in line with our promise of being “Committed to life” – we improve the life of every single person.
Their goal for patients is to receive optimal care and regain a sense of normality
Fresenius Kabi aims to support patients throughout their entire cancer treatment. Their goal for patients is to receive optimal care and regain a sense of normality to the greatest extent possible. The portfolio at their disposal ranges from pharmaceuticals to medical clinical nutrition products and other support services.
Highly effective medicines, such as a wide range of oncology products as chemotherapeutic drugs and biosimilars, can be used today to personalize therapies. Generics and biosimilars are not only used to fight tumors – some also aid in reducing side effects and improving quality of life.
Cancer and its burdensome treatment can impair the nutritional status of patients and significantly weaken the body. Medical nutrition products for cancer patients support in counteracting malnutrition and help in providing patients with the key nutrients needed during critical phases of their disease.
Fighting this disease remains an exceptional situation for the patients and their loved ones, during which they should not be left on their own. That is why Fresenius Kabi provides education and support programs that benefit mental well-being throughout the entire treatment journey, for example, during their transition from hospital treatment to home care.

Long-term aftercare also plays an important role where children are concerned
Helios relies on holistic oncology in the treatment of cancer. It takes into account the individual person and their medical history and ensures that each patient receives the best individual treatment.
Cancer is and remains a difficult time in someone’s life – especially if that person develops it very early. Sick children have particular needs, and pediatric oncology staff across four Helios Hospitals in Germany are committed to meeting them.
One of their youngest patients is Mika, a boy who was diagnosed with acute lymphoblastic leukemia in 2024 at just four years old. At the Helios Hospitals in Schwerin, he receives cutting-edge chemotherapy tailored to his young age. Although he has a 90 percent chance of being cured, the treatment is an enormous burden for the boy and his family, both physically and mentally, which is why the employees also offer them psychological support. Long-term aftercare also plays an important role where children are concerned in order to prevent late effects. At Helios, for example, onco-psychologists are always involved in the therapy process.
Individual cancer treatment and support is of crucial importance, regardless of the patient’s age. To this end, specialists across different disciplines work closely together at Helios to find the best combination of diagnostics, personalized therapy, and aftercare.
Quirónsalud focuses even more on people when providing care
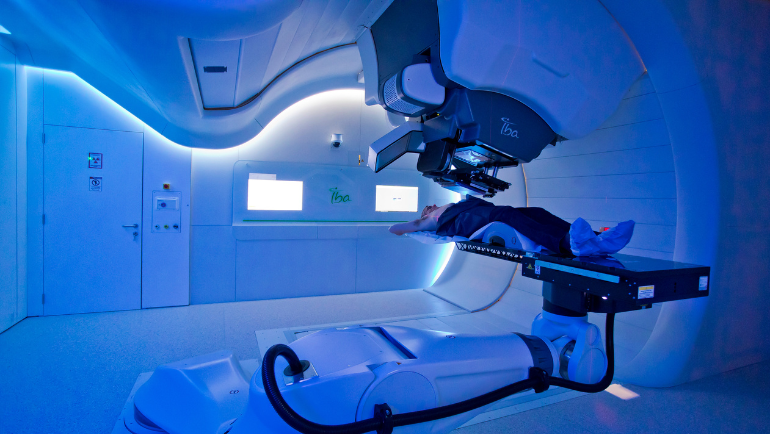
Patients treated by Quirónsalud in Spain also benefit from a holistic approach and innovative therapy methods. The hospital group is part of Fresenius Helios and has oncology departments staffed by highly specialized professionals in almost all hospitals. They work closely with other teams in multidisciplinary committees and use state-of-the-art technology to diagnose and treat a wide range of tumors. By way of example, Quirónsalud is also one of the pioneers in proton therapy, a form of radiation treatment that targets tumors with millimeter precision.
Doctors and nursing staff are always close to the patient, i.e. ‘human to human’. In order to focus even more on people when providing care, the Jiménez Díaz Foundation in Madrid, for example, has launched a project named HOPE in its oncology day clinic. The patient-centered initiative uses digitalization to help people avoid waiting times and thus gain valuable time. Since the launch of the tool, everyone involved has been less stressed, while the patients’ quality of life has markedly improved. It has also increased the efficacy and safety of the therapies.
Innovative therapies can save lives
Wherever Fresenius’ medical teams operate today, they do their best to ensure that every patient gets access to the best possible medical care. Patients like Mika in Schwerin, who is by no means out of the woods just yet, but is now getting better.
Mika’s father started a blog during his treatment to help him cope better during this difficult time – generating a huge wave of public sympathy. As a sign of gratitude, his father turned this attention to account for a good cause, raising more than 30,000 euros to date for the children’s cancer charity Kinderkrebshilfe in Germany and cancer research at Helios in Schwerin.
After all, this is what it is all about. Innovative therapies, which are rapidly being developed thanks to modern research, can ultimately save lives. Experts at Helios expect important new milestones to be reached in cancer therapy over the next few years. Hopes are pinned, for example, on CAR-T cell therapies, highly effective antibody-drug conjugates, and tumor vaccines, which are special vaccinations that prevent cancer from developing in the first place. Therapy and prevention will keep on improving – so that people can continue to move out of the shadow cast by the disease in the fight against cancer.
Learn more about new cancer therapies
CAR T cell therapy: Innovative therapy with the aid of genetically modified cel… The new ADC medicines represent a real revolution in treatment HOPE gives hopeA consulting room on a sunny morning in January at Madrid’s Villalba University Hospital, part of the Quirónsalud Hospital Group. The patient is visibly nervous. Her head is full of questions and diffuse fears – which is not uncommon. Dr. Natalia Ruiz Micó, Head of the Orthopedic Surgery and Traumatology Department, tries to reassure her. “Today we are going to talk about your upcoming hip operation …” That is how the doctor begins her consultation with the patient. This is also not uncommon. It is a conversation that will be held thousands of times a day here and at other hospitals. One thing, however, is different: Dr. Ruiz can fully concentrate on her patient. She doesn’t have to study the long medical records or take any notes, which means that she is less stressed than she used to be. That’s because these tasks are now handled by Scribe, an intelligent AI-based dictating machine for medical purposes that she opens on her work phone.

(Published: June 2025)
Scribe was launched in the summer of last year. To date, more than 4,000 doctors in a total of 50 Quirónsalud hospitals across Spain have already used it to carry out over 500,000 consultations. Both doctors and patients report positive experiences of the technology.
Among the first users was the Department of Orthopedic Surgery and Traumatology at Madrid’s Villalba University Hospital, where Dr. Ruiz is chief physician. While she talks to her patient, Scribe records the conversation, transcribes it, and summarizes the essential points and the resulting tasks. If necessary, the tool can even switch back and forth between several languages.
The task of clinical documentation was previously a tedious job that was shared among the hospital teams. The secretary’s office took on some of it, as did the assistant doctors on the ward, but on many an occasion the team of treating doctors had to do it themselves. Scribe now saves everyone on the team a great deal of time – or to be more precise, the time previously spent on documentation and administrative work during each consultation. “Medical documentation is a very important aspect of our work, but it is also very time-consuming. It has been difficult to automate until now because it requires special knowledge,” says Dr. Ruiz.

Technology with huge potential
Why is this now possible with Scribe? As is so often the case, the answer is AI, or more precisely, generative artificial intelligence, which has been enjoying a worldwide boom since the launch of ChatGPT. This type of AI can reliably process human speech and generate high-quality speech output in real time. Yet Scribe isn’t a product of Silicon Valley. Quirónsalud developed the AI solution itself in conjunction with an IT partner and has been testing it since June 2024.
“This technology has huge potential – and not just for Quirónsalud. Scribe helps to humanize healthcare and establish a much closer doctor–patient relationship. It frees medical teams from their administrative tasks and allows them to focus more on their patients,” says Dr. Jorge Short Apellániz, Deputy Medical Director of the Fundación Jiménez Díaz University Hospital in Madrid and one of the initiators of this project at Quirónsalud.
Yet the tool doesn’t just save time. It also guides the doctor independently through the consultation. “Based on the information collected, Scribe also suggests necessary diagnostic tests or therapy options,” explains Dr. Emilio Calvo, Head of Orthopedic Surgery and Traumatology at the Fundación Jiménez Díaz University Hospital in Madrid. “So even during a consultation, the tool highlights possible courses of action,” says Calvo. He stresses, however, that the final decision is still made by the doctor.

Part of an intelligent clinic network
In order for Scribe to be able to make such intelligent suggestions, it has not only been specially trained, but is also embedded in an overarching digital system developed by Quirónsalud: the Casiopea hospital portal (see box for more information). All information is consolidated here, including the digital medical records – provided that the patients have consented to this. Casiopea has also supplied key training data for Scribe. “This high-quality and very specific data ensures that Scribe does not invent information, but instead delivers relevant results,” explains Dr. Short Apellániz. Furthermore, IT security and data protection are of the utmost importance where sensitive data of this nature is involved.
And how have patients reacted so far to the new development? Dr. Carolina Gotera, senior pulmonology physician at the Fundación Jiménez Díaz University Hospital, gets a lot of positive feedback when she explains the program: “Most patients congratulate us because they recognize the potential of AI and can see how innovative we are. They think it is good that we have more time for them, and their comments are therefore very positive.”
Digital pioneer in Europe
With Scribe, Quirónsalud is continuing to consolidate its position as a digital pioneer in Europe – demonstrating how innovations in healthcare directly benefit people. “The tool improves communication between patients and medical staff, while minimizing any errors and misunderstandings,” summarizes Dr. Short. “At the same time, such tools increase efficiency and help to reduce the administrative burden for our employees. We can then all invest the time saved in interacting with the patients.” Quirónsalud plans to introduce the Scribe tool in all medical service areas and departments at its various hospitals across Spain in the course of 2025.
"It's a game changer"
For the development of Scribe, the Quirónsalud Operations Team was honored with the Else Kröner Award 2025. This internal award recognizes Fresenius teams for outstanding, innovative, and exceptional initiatives that bring the Fresenius Principles to life and carry on Else Kröner’s legacy.
Discover the people and ideas behind the innovation - watch the full interview now.
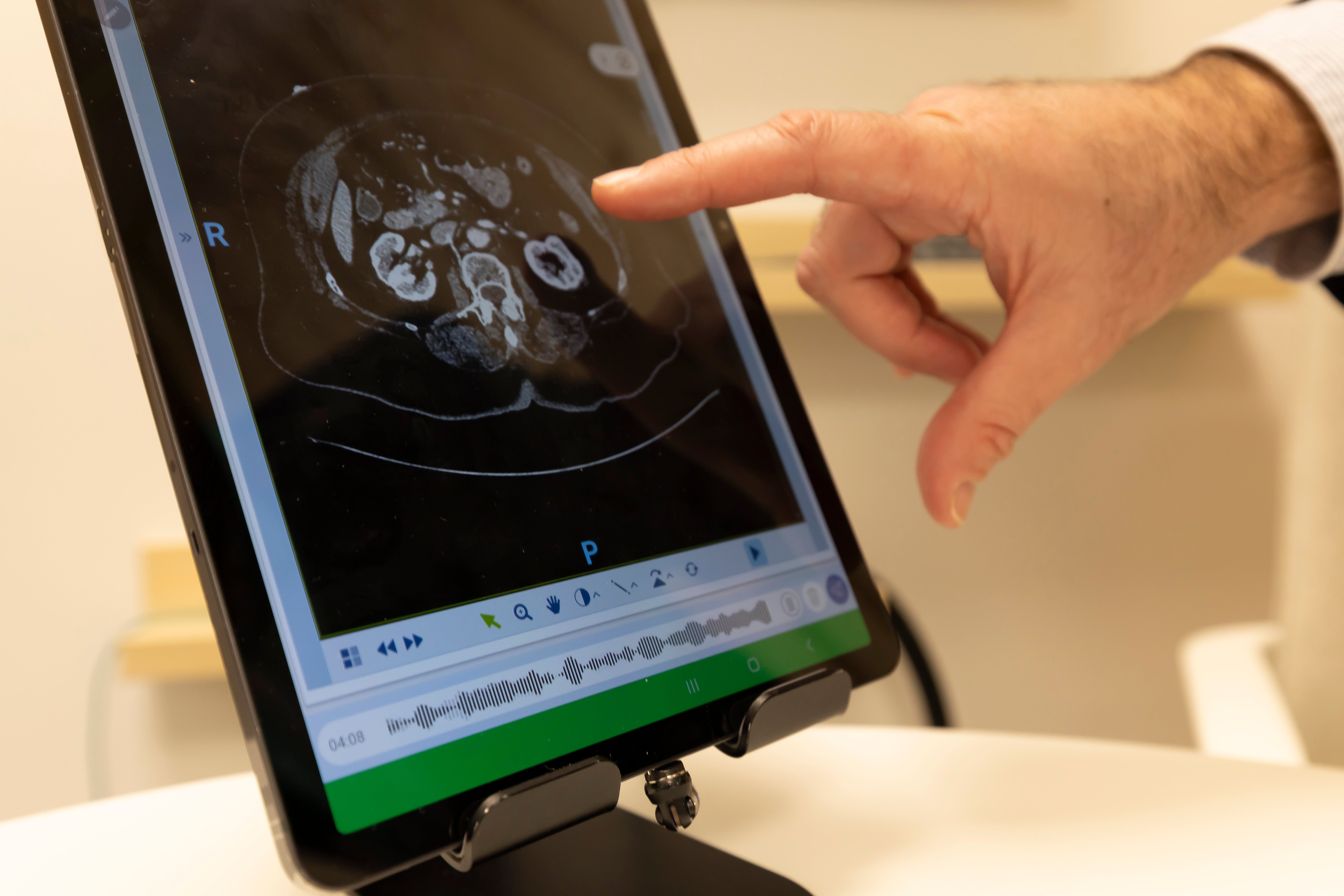
Casiopea – at the heart of the digital hospital
The Casiopea hospital portal – a so-called digital ecosystem – is a platform that Quirónsalud has been using throughout Spain since 2020. It optimizes patient flows and seamlessly connects all stages of the treatment process – from diagnosis and therapy to outpatient care. Almost eight million users in Spain are already connected to the system via a patient app. Doctors also use the Casiopea Mobility app on their smartphones, making it possible to arrange remote consultations, view laboratory results, and issue prescriptions, among many other things.
Fresenius is currently examining how the digital Casiopea platform can be transferred to Helios hospitals in Germany as part of a pilot project, taking into account the specific requirements of the German market. Among other things, it is working on a central communication interface between the doctor and patient.
Learn more about AI at Fresenius
Fresenius Sustainability Highlights Magazine 2024
Antibiotics are medicines that treat and prevent various bacterial infections in humans and are a mainstay of modern medicine. But they also have their downsides: Antimicrobial resistance, often abbreviated as AMR, is on the rise worldwide. This includes resistance to other medicines used to fight infections caused by fungi or viruses.
(Published: November 2025)
The main cause of AMR is the misuse and overuse of antibiotics / antimicrobial treatments in humans, animals and plants, leading to the development of drug-resistant pathogens.
Those involved in the healthcare system thus hold a particularly high level of responsibility: that includes doctors, nurses, pharmacies – but also pharmaceutical manufacturers, and not least the patients themselves. In this context, taking responsibility not only means ensuring prudent administration and intake of antibiotic medicines, but also responsible manufacturing including controlled production.
We are committed to minimizing the environmental impact of antibiotic production at all sites!
Prevention is a key element in production for Fresenius Kabi. As a leading manufacturer of intravenous antibiotics, the business sector has a dual responsibility: on the one hand, to produce essential medicines and make them available worldwide, and on the other hand, to ensure responsible manufacturing. By applying the standard, we ensure proper control of waste and wastewater so that any antibiotic residues are minimized, controlled, and effectively treated, thereby reducing environmental risks and minimizing the potential for antimicrobial resistance.
Careful handling of antibiotic residues is particularly important: If they enter the environment via wastewater, they can contribute to the development of resistant pathogens there. “To counteract and prevent this, we are committed to minimizing environmental impact in antibiotic production at all our sites,” says Kirsten Nyland, Senior Vice President Environmental & Occupational Management at Fresenius Kabi. Since 2020, Fresenius Kabi has been a member of the Antimicrobial Resistance Industry Alliance (AMRIA) and is working together with other companies on solutions. “Because: How we handle these resources today will determine whether antibiotics will still be effective tomorrow,” Nyland adds.
A flagship project has been implemented at the site in Santiago de Besteiros, Portugal: It became the first Fresenius Kabi site to receive BSI Kitemark™ certification for minimized AMR risk in the production of ceftriaxone. The certification confirms that the manufacturing processes there comply with the “AMR Industry Alliance Antibiotic Manufacturing Standard”. Recently, the Fresenius Kabi site in Kutno, Poland, also received the certification to produce amikacin and linezolid. At the production unit in Graz, Austria, AMR certification was a natural next step. The site had long adhered to stringent environmental and quality standards, making BSI Kitemark™ certification for minimized AMR risk in the production of amikacin a confirmation of existing best practices. The certifications also recognize the sites’ environmental management – in particular the measures taken to control and reduce antibiotic residues in wastewater and waste streams. Fresenius Kabi is thus working on an ongoing basis to make a measurable contribution to the containment of AMR.

Why Helios and Quirónsalud have established consistent antibiotic management
Helios and Quirónsalud use antibiotics to treat bacterial infections in their hospitals. That is precisely why it is crucial to prevent the emergence of resistant pathogens at an early stage, because AMR in hospitals can prolong the length of stay, increase the risk of complications, and influence mortality.
Helios is actively taking on this challenge: “In order to detect abnormalities and possible incorrect use of antibiotics, their consumption needs to be monitored. That is why we introduced antibiotic reporting in all Helios hospitals as long ago as 2012,” says Dr. Felix Giebel, Chief Physician in the Department of Infection Prevention and Hospital Hygiene and Head of the Infectious Diseases Division. “The burden of antimicrobial resistance is already high and may continue to increase – meaning even fewer antibiotics could be effective in the future. Only targeted antibiotic management can prevent or at least curb that.”
Helios and Quirónsalud rely on interdisciplinary teams and digital tools
Effective hygiene measures are a key lever for preventing AMR – with the aim of avoiding nosocomial infections, in other words, those acquired during a stay in hospital. Helios goes even further:
Helios introduced a holistic antibiotic stewardship program in 2019. It includes certified digital training of antibiotic stewardship experts (ABS), the implementation of ABS-trained physicians in every clinical unit, the introduction of a digitally controlled prescription of antibiotics, and a central infection monitoring system, referred to as “Surveillance”. Practical measures and guidelines for the hospitals are derived from this program on an ongoing basis.
Quirónsalud hospitals also provide examples of their firm commitment to research and sustainability. In 2020, Quirónsalud Barcelona Hospital launched the “PROA” programme, in Spanish “Programa de Optimización del Uso de Antimicrobianos”. It promotes targeted and rational antibiotic use based on standardized guidelines, interdisciplinary teams, and ongoing monitoring of prescriptions, and is firmly embedded in the hospital’s quality and infection-prevention policies. Another example is the ARGOS project, developed by Quirónsalud colleagues. It is an integrated infection control and prevention program, using evidence, collaboration, and AI to guide safer antibiotic use.
Further information
AMR occurs when microorganisms have changed in such a way that they no longer respond to antibiotics, i.e. they have become resistant. If such resistant pathogens enter the environment, they can spread infections and make diseases that were previously easily treatable become life-threatening. AMR thus affects us all. The WHO has warned that without countermeasures, up to 10 million people a year could die from the consequences by 2050.

Surgery to resolve a life-threatening diaphragmatic hernia has successfully been performed on a preterm baby weighing little more than 500 grams for the first time ever – an unprecedented procedure and medical breakthrough that brings hope.
(Published: November 2025)
Born three months preterm weighing just 490 grams, the little girl was barely bigger than an adult’s hand. Doctors delivered her by C-section on January 23, 2025 after her mother developed pregnancy-related complications. Senior Consultant in Obstetrics and Gynecology Dr. Friederike Senger discovered during an ultrasound before the little girl had even taken her first breath that she had a life-threatening defect. In this condition known as a diaphragmatic hernia, the abdominal organs move up into the chest cavity. This makes breathing almost impossible and would previously have been considered inoperable in such a tiny infant.
“Based on this diagnosis, we had to tell the girl’s parents that their daughter had only a very slim chance of surviving. I assured them, though, that we would do everything medically possible to try to save their tiny newborn,” recalls Dr. Kai Siedler, Chief Physician for Pediatrics and Adolescent Medicine at the Helios hospital in Pforzheim.
An interdisciplinary team from two Helios sites – and a strong will to live
Thanks to highly specialized care with state-of-the-art ventilation technology and special feeding strategies, the neonatal team led by Chief Physician Dr. Kai Siedler and Senior Physician Alexandra Weber successfully stabilized the little girl, saving her life. In the days that followed, they were even able to start giving her breast milk, drop by drop via a tiny feeding tube.
An interdisciplinary team of surgeons and pediatricians from two Helios sites operated on the frail little girl when she was just 19 days old and weighed only 540 grams. Chief Physician Dr. Thomas Ringle and Senior Physician Max Böttinger-Hummel from the pediatric surgery department at Helios Klinikum Pforzheim and Prof. Dr. Stefan Gfrörer, Chief Physician for Pediatric Surgery at Helios Klinikum Berlin-Buch used special magnifying loupes to perform the procedure. They successfully closed the hole in the diaphragm and returned the organs to their correct position – in a newborn whose organs had only just begun to develop.
“We broke new ground with this surgery at such an extremely low birth weight. Without it, the child would have had no chance of surviving. The perfect cooperation between our experienced interdisciplinary team of surgeons, pediatric intensive care physicians, and intensive care nurses was key to its success. Our little patient’s sheer determination to live also impressed us time and again though,” emphasizes Dr. Thomas Ringle.
Aftercare by a highly committed team of nurses
After the successful surgery, the little girl received round-the-clock care from the dedicated nursing team in the pediatric intensive care unit, then on the intermediate care ward. She made excellent progress: There were no complications, her breathing stabilized, she gained weight, and began drinking on her own.
On 3 June, 130 days after her birth, the little patient could be discharged from hospital weighing 2,150 grams. She will continue to be treated as an outpatient and will return to the children’s hospital for any procedures she may need in the future.
Dr. Kai Siedler hails the surgery as a milestone: “This case shows what modern medicine can achieve today – but above all what is possible when a highly motivated team works together. We are proud of everyone involved.”
Despite their tremendous concern, the baby’s parents were always optimistic: “We were terrified that we might lose our child, but we never lost hope. We owe the fact that our daughter is alive today and thriving like she is to the courage and skill of the physicians and the entire pediatric care team. We will forever be grateful that we chose this hospital.”
This success once again highlights the outstanding expertise and exceptional teamwork of the Fresenius Helios staff in caring for high-risk preterm infants. And it should set a precedent: The experience gained from this procedure will be shared with the international medical community.
What does it truly mean to live with a chronic illness? Beyond medical symptoms, patients face daily challenges that impact their routines, emotions, and interactions with the healthcare system. To foster a deeper understanding of these realities, Fresenius Kabi is participating in an immersive patient experience program, allowing colleagues to step into the world of those they serve.
(Published: July 2025)
Employees partake in a simulation that replicates certain situations in the life of a patient. Using a dedicated app, they receive instructions and updates, engage in role-play calls with actors portraying caregivers and healthcare professionals, and interact with physical materials received in advance to go through practical aspects of managing symptoms. For 24 hours, they are not just employees working in healthcare. They become patients. This hands-on experience provides valuable insights – not only into the physical symptoms of a disease but also into the emotional and logistical burdens that come with managing a chronic condition.
Transforming empathy into action
To bring this immersive experience to life, Fresenius Kabi is partnering with A Life in a Day, an award-winning program designed to offer a realistic glimpse into the daily challenges of living with a chronic illness. Developed in close collaboration with patients, patient support organizations, and healthcare professionals, the program ensures accuracy and authenticity.
For our people, it has already made a lasting impression. In the most recent wave of the program, colleagues took part in the Crohn’s disease simulation, a condition that causes chronic inflammation of the gastrointestinal tract. Crohn’s can lead to severe abdominal pain, diarrhea, fatigue, and weight loss, but its impact extends beyond physical symptoms. Patients must navigate complex treatment regimens, frequent medical appointments, and the unpredictability of flare-ups which can lead to distressing situations and difficult choices about daily activities – all while balancing everyday life. Through this experience, participants gained a first-hand perspective on these challenges, strengthening their understanding of patient needs and the full burden of their disease.
Improving healthcare by understanding the patient perspective
This initiative aligns closely with the Fresenius principles, particularly our commitment to patient-centric care. It reminds us that healthcare is, at its core, human to human.
By fostering deeper empathy and insight, it empowers colleagues to serve patients beyond expectations and develop solutions that truly address their daily struggles. Whether in direct patient care or the development of innovative therapies, understanding the patient perspective is essential to improving healthcare. Committed to Life, the Fresenius promise, reflects this belief.

Nutrition is a topic that surrounds us every day. While we often talk about obesity and weight loss diets, we actually overlook that the opposite, meaning malnutrition or undernutrition, can be as detrimental. We speak of malnutrition when the body is not provided with enough nutrients, such as protein, carbohydrates, fats, vitamins, and minerals, which are essential for maintaining all vital body functions.
When our nutritional status is not optimal, the body becomes less resilient in facing challenges like illness, colds, or infections. Improving our nutritional status will therefore support and protect our body.

(Published: November 2025)
50 Years of Fresubin
This year marks the 50th anniversary of Fresubin®, Fresenius Kabi’s trusted enteral nutrition brand. Since 1975, Fresubin® has supported over 300 million people in more than 75 countries, helping patients with their dietary management and therefore connection to life when everyday nourishment becomes a challenge.
From chronic disease and critical illness to aging, cancer care, and rehabilitation, Fresubin® has played a vital role in restoring nutritional support. Because when eating becomes difficult or impossible, nutrition is no longer just fuel. It becomes a foundation of patients’ life.
- Find out more about medical nutrition.
- Find out more about Fresubin on fresubin.com or read our the full "50 years of Fresubin" story on fresenius-kabi.com

(Published: December 2025)
Concentration fills the operating room, high-resolution images can be seen on several screens, the camera view penetrates deep into the finest branches of the bronchial tree, while a so-called digital twin – the personalized 3D plan of a lung – provides guidance. Dr. Joanna Krist, Senior Pulmonary Physician in Heckeshorn Lung Clinic at Helios Hospital Emil von Behring in Berlin, is looking at her control panel. With a steady hand, she guides an ultrathin, flexible catheter deep into the airways of her 59-year-old patient. He has been a heavy smoker all his life, now there is a suspicion of lung cancer. Krist follows the virtual path of the catheter on the central navigation monitor – bright blue glowing lines show her the course to the destination point: a tiny shadow in the lung tissue. A small tissue sample, which she will take shortly, should now provide certainty.
What sounds like a high-tech future has long been reality: Doctors at the highly specialized Helios lung cancer centers in Berlin and Wiesbaden are already setting new standards in the early diagnosis of disease with the robot-assisted “Ion endoluminal system” for bronchoscopy.
What you should know about lung cancer
Lung cancer remains the most common cause of cancer-related deaths in Germany and one of the most dangerous oncological diseases of all. Around 45,000 people in the country die of it every year – because the disease is often only diagnosed at an advanced stage. “The problem is that in the early stages, lung cancer causes hardly any symptoms or even discomfort, so it is usually discovered late – often when metastases are already present and treatment is no longer possible,” explains Prof. Dr. Tim Hirche, Director of the Clinic for Pulmonology at Helios HSK Wiesbaden.
The Ion system marks a turning point here. “The new robotic system for bronchoscopy is a game changer,” says Dr. Krist. “We can use it to get to the deepest lung tissue and can even reach millimeter-sized suspicious nodules – so-called coin lesions – and take samples. This has not been possible with endoscopy until now.”
Her colleague, Prof. Hirche, adds: “Ion enables us to give our patients certainty much earlier – and thus often offer them the chance of treatment. It is a real paradigm shift in the field of pulmonology.” The Ion system combines robotics, imaging techniques, and interdisciplinary medical expertise in a hitherto unparalleled manner. Find out more in the box.
Certified by the German Cancer Society, the Helios lung cancer centers in Berlin and Wiesbaden are among the first hospitals in Germany to use the robotic system for regular patient care. The technology also has scientific support at both Helios sites, with patients suitable for the procedure also being able to participate in studies.

From diagnosis to therapy
If the suspicion of lung cancer is confirmed after the examination, an interdisciplinary network immediately comes into play at Helios. Lung specialists, thoracic surgeons, oncologists, and radiologists get together on the tumor board to discuss the individual treatment plan. “Depending on the stage, this treatment can range from minimally invasive surgery in one of our two robotics centers to radiation therapy and chemotherapy or immunotherapy,” says Prof. Dr. Torsten Bauer, Chief Pulmonology Physician in Heckeshorn Lung Clinic at Helios Hospital Emil von Behring.
“The precise diagnostics using the robotic system enables us to start treatment much earlier,” stresses the specialist. With the new procedure, physicians are moving a step closer to the goal of diagnosing lung cancer earlier, less invasively, and more precisely – before symptoms even appear. For patients, this means a much lower risk, less stress, less uncertainty – and, ideally, an opportunity to gain crucial time for early treatment.
Further information
The Ion endoluminal system is a robot-assisted bronchoscopy system that allows physicians to safely reach even tiny changes in the lung tissue. Before the actual procedure – bronchoscopy – the planning software creates a detailed 3D model – the digital twin – on the basis of the CT scan carried out in advance. This makes it possible to work out the optimal pathway to the suspicious site – in a similar way to a GPS or a navigation system.
During the examination, which is carried out under anesthesia, the highly flexible catheter, equipped with sensors and a camera, is inserted into the bronchi via a breathing tube and guided with millimeter precision. At the same time, the medical practitioners also check its position with the help of state-of-the-art scanning technology – a so-called cone beam CT. Thanks to precise real-time imaging, even the smallest coin lesions can be reached with pinpoint accuracy. Tissue samples can then be taken and analyzed in a procedure that is less invasive for the patient, with a lower risk of complications than with previous procedures.

#FutureFresenius is making the company fit for the future: Fresenius is focussing on its Operating Companies Fresenius Kabi and Fresenius Helios, so on the three therapy platforms (Bio)Pharma, MedTech and Care Provision. They cater to system-critical areas of healthcare and are geared toward value creation and profitable growth. The two Operating Companies are to now also work together more closely. And the brand identity must reflect and support this.
(Published: May 2024)
Fresenius CEO Michael Sen introduced the new brand identity to the public at the Annual General Meeting in May 2024. It will now be rolled out across the Group by the end of the year.
“Fresenius is now fresher and more modern. The company logo has not been changed entirely, but rather revamped. Among others, we have updated the color palette to fit perfectly with the digital world. This brand image stands for a new Fresenius. Internally, we hope that the uniform brand identity will help us to grow even closer together as Team Fresenius. Because regardless where we work, we are all Fresenius. Externally, we want Fresenius to be recognized as a leading global healthcare company. A brand is always a promise. For us, our promise is “Committed to Life,” whereby we consistently strive to improve people’s lives”, said Michael Sen, CEO of Fresenius.

A person is more than their illness
With Fresenius Kabi, Helios and, Quirónsalud, the company has a unique portfolio and expertise. It understands the life dimensions of illness and health better than any pure medical technology or pharmaceutical company ever could. The new brand identity aims to support exactly this: The new Fresenius. Because regardless of where employees work, they are all Fresenius! Externally, the company wants to create a consistent image and to be recognized as a leading global healthcare company. Which is why Fresenius has revamped all aspects of the brand image: the logos, colors and shapes, fonts and images. Particularly the bright, modern color palette perfectly fits into the digital world. The Kabi, Helios, and Quirónsalud logos will remain. However, they will be incorporated into the new Fresenius world visually so that it is always clear that Fresenius is ONE team.
Fresenius sees that people are more than just their illness. It sees the lives they lead – within their families, their communities, and their jobs. It accompanies people throughout their whole lives, for better or for worse. Fresenius is there when it matters. This is what the entire #TeamFresenius works toward each and every day with its 190,000 colleagues.
A brand is always a promise; for Fresenius, a promise to its customers – and especially its patients. The Fresenius promise is “Committed to Life.” The company consistently strives to improve people’s lives.

Key brand elements: more than the sum of its parts
Logo
The new Fresenius logo expresses the company’s self-image as a symbol and word mark. The geometric lines of the existing logo have been replaced with more emotional and organic forms. At the same time, its shape reminds of the “F” in Fresenius and brings the company’s brands together with a new common color scheme.

“Bond for Life”
One key design element is the “Bond for Life,” which derives from the lifelines of humans. It symbolizes the different needs that humans have during various phases in their lives. The “Bond for Life” is inspired by the lifeline in the palm of our hands and signifies people’s needs during the different phases in their lives – with all their ups and downs. These reflect the connection between all of the Fresenius businesses, which support people at every stage in their lives as best possible.

Rollout by the end of the year
Fresenius has planned the implementation of the relaunch in phases: The first phase is completed with the Annual General Meeting and introduction of the new Fresenius brand at corporate level.
Now it's the Operating Companies' turn next: Their appearances will be harmonized with the new Fresenius brand presence and drive the launch forward together. Cross-business teams are already working together here. There will also be information and guidelines on this in the coming weeks - until then, the previous design of the Operating Companies will apply.
The final harmonization and completed new brand architecture should be jointly defined and introduced by the end of the year.







